Inspiratory Flow Profiles
In a clinical study,
Patients with moderate to severe CF were able to use the PODHALER device1
Patient types that were able to generate the inspiratory flow rates and volumes required to receive medication included1
- Older patients with significant disease progression and associated decreases in FEV1
- Younger patients (aged >6 years) with inhaled volumes <1 L
Patients followed the Instructions for Use. Pediatric patients aged 6 to 10 years of age with FEV1 <40% predicted were not evaluated.
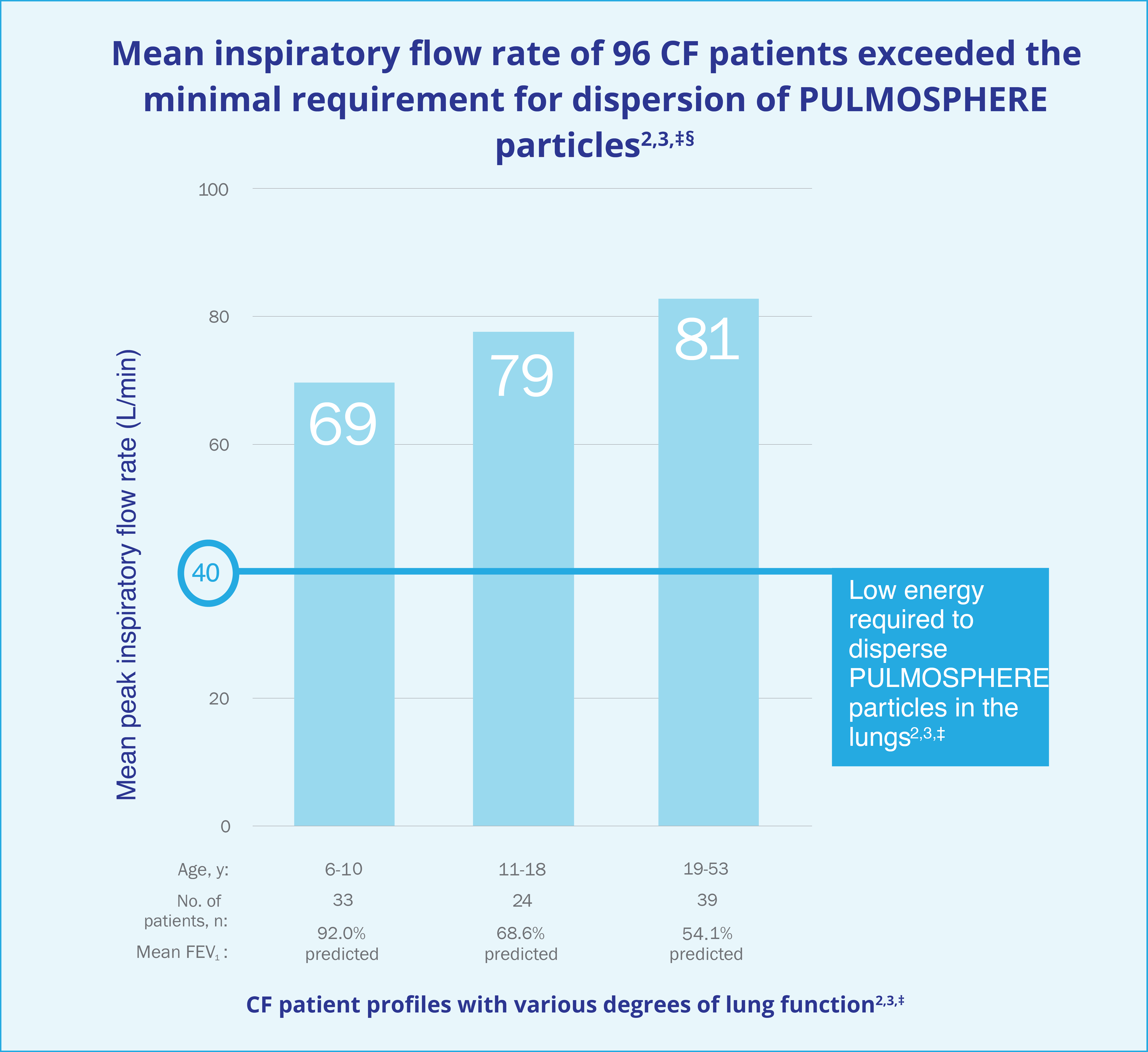
§A flow rate of 40 L/min represents a flow rate more than 2 standard deviations below the mean peak inspiratory flow rates measured for pediatric patients in the study.4
References
- TOBI® PODHALER® [Prescribing Information, 2023].
- Tiddens HA, Geller DE, Challoner P, et al. Effect of dry powder inhaler resistance on the inspiratory flow rates and volumes of cystic fibrosis patients of six years and older. J Aerosol Med. 2006;19(4):456-465.
- Data on file [TSB-001 The inspiratory volume and flow of cystic fibrosis patients while using a simulated dry powder inhaler]. Mylan Specialty L.P.; 2003.
- Geller DE, Weers J, Heuerding S. Development of an inhaled dry-powder formulation of tobramycin using PulmoSphere® Technology. J Aerosol Med Pulm Drug Deliv. 2011;24(4):175-182.

 MORE +
LESS –
MORE +
LESS –
INDICATION
TOBI® PODHALER® (Tobramycin Inhalation Powder) 28 mg per capsule is indicated for the management of cystic fibrosis patients with Pseudomonas aeruginosa.
Safety and efficacy have not been demonstrated in patients under the age of 6 years, patients with forced expiratory volume in 1 second (FEV1) <25% or >80% predicted, or patients colonized with Burkholderia cepacia.
IMPORTANT SAFETY INFORMATION
TOBI PODHALER is contraindicated in patients with known hypersensitivity to any aminoglycoside.
Bronchospasm can occur with inhalation of TOBI PODHALER. Bronchospasm should be treated as medically appropriate.
Caution should be exercised when prescribing TOBI PODHALER to patients with known or suspected auditory, vestibular, renal, or neuromuscular dysfunction.
Ototoxicity, as measured by complaints of hearing loss or tinnitus, was reported by patients in the TOBI PODHALER clinical studies. Tinnitus may be a sentinel symptom of ototoxicity, and therefore the onset of this symptom warrants caution. Ototoxicity, manifested as both auditory and vestibular toxicity, has been reported with parenteral aminoglycosides. Vestibular toxicity may be manifested by vertigo, ataxia, or dizziness.
Cases of ototoxicity with aminoglycosides have been observed in patients with certain variants in the mitochondrially encoded 12S rRNA gene (MT-RNR1), particularly the m.1555A>G variant. Ototoxicity occurred in some patients even when their aminoglycoside serum levels were within the recommended range. Mitochondrial DNA variants are present in less than 1% of the general US population, and the proportion of the variant carriers who may develop ototoxicity as well as the severity of ototoxicity is unknown. In case of known maternal history of ototoxicity due to aminoglycoside use or a known mitochondrial DNA variant in the patient, consider alternative treatments other than aminoglycosides unless the increased risk of permanent hearing loss is outweighed by the severity of infection and lack of safe and effective alternative therapies.
Caution should be exercised when prescribing TOBI PODHALER to patients with known or suspected renal dysfunction. Nephrotoxicity was not observed during TOBI PODHALER clinical studies but has been associated with aminoglycosides as a class.
TOBI PODHALER should be used cautiously in patients with neuromuscular disorders, such as myasthenia gravis or Parkinson’s disease, since aminoglycosides may aggravate muscle weakness because of a potential curare-like effect on neuromuscular function.
Aminoglycosides can cause fetal harm when administered to a pregnant woman. Patients who use TOBI PODHALER during pregnancy, or who become pregnant while taking TOBI PODHALER, should be apprised of the potential hazard to the fetus. The amount of tobramycin excreted in human breast milk is unknown. However, systemic absorption of tobramycin following inhaled administration is expected to be minimal. A decision should be made whether to discontinue nursing or TOBI PODHALER. TOBI may cause intestinal flora alteration. Advise a woman to monitor the breastfed infant for loose or bloody stools and candidiasis.
Patients receiving concomitant TOBI and parenteral aminoglycoside therapy should be monitored as clinically appropriate for toxicities associated with aminoglycosides as a class. Serum tobramycin levels should be monitored.
Concurrent and/or sequential use of TOBI PODHALER with other drugs with neurotoxic, nephrotoxic, or ototoxic potential should be avoided. Some diuretics can enhance aminoglycoside toxicity by altering antibiotic concentrations in serum and tissue. TOBI PODHALER should not be administered concomitantly with ethacrynic acid, furosemide, urea, or mannitol.
In a clinical trial, the most commonly observed adverse events with TOBI PODHALER occurring at a frequency of at least 10%, were cough, lung disorder, productive cough, dyspnea, pyrexia, oropharyngeal pain, dysphonia, hemoptysis, and headache.
Please see Full Prescribing Information

